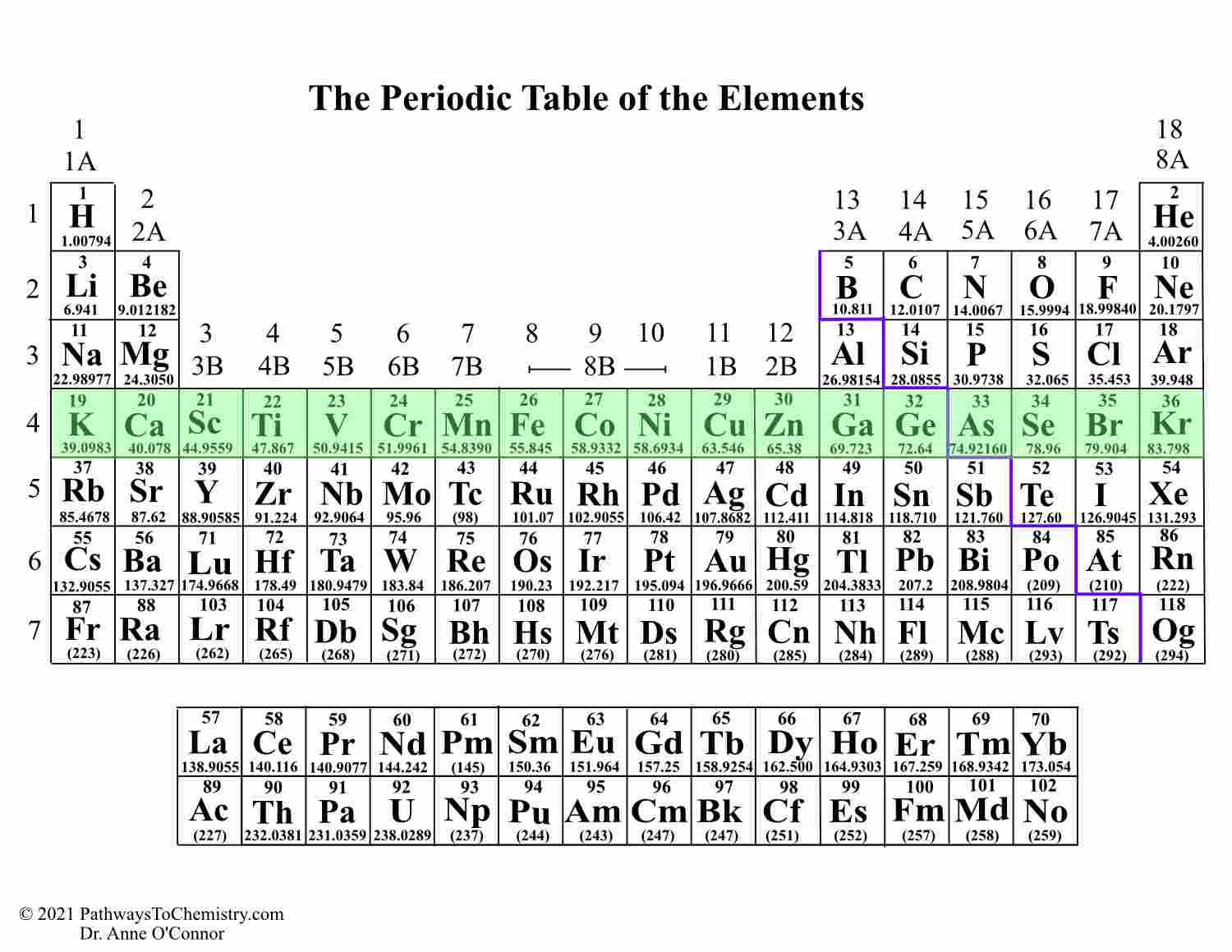
The periodic table as we use it today was created by Dmitri Mendeleev in 1869. When was the Periodic Table of Elements Created? Some of these groups have quite different properties from others, like the noble gases, but some share similar properties, like the transition metals, and are sometimes considered together. There are 18 groups in the periodic table, corresponding to its 18 columns and they are numbered from left to right. Each group contains elements with similar physical or chemical characteristics, which mostly arise from the number of electrons in their outermost electron shell (called the valence shell). This is why each column ends up containing elements that have similar properties – each column is called a group.Ī group is a column of elements in the periodic table. Once the end of a period is reached, the elements continue in the next period below, such that they end up underneath an element with similar properties. The periodic table is arranged to recognise that certain chemical and physical properties of the elements recur – this is called the Periodic Law. The properties also change across a period – that’s why metals are on the left, and non-metals on the right.Īs the name suggests, the periodic table is periodic – something recurs after a given interval. The elements are arranged in order of increasing atomic number across a period from left to right, so each element has one more proton in its nucleus compared to the preceding one. (You can think of an electron shell as an orbit that an electron follows around the nucleus of the atom.) Each element in a period has the name number of electron shells. The rows of the periodic table are called periods. Research into new elements is an ongoing process. The discovery and naming of elements is now overseen by the International Union of Pure and Applied Chemistry (IUPAC). A few elements are found in a pure form, like gold. Most substances we find on earth are composed of compounds, which are combinations of elements. For example, atoms with just one proton are hydrogen, atoms with two protons are helium, atoms with six protons are carbon, and so on. We use the number of protons in the nucleus to identify the element, and call this its atomic number ( Z). The term element refers to atoms that have a specific number of protons (a subatomic particle with a positive charge +) in their atomic nuclei. The periodic table is an attempt to do this by sorting elements into periods and groups according to the number of electron shells and their electronic configuration. As there are 118 known elements (at this point), it is important that they be organised in a convenient and sensible way. The periodic table of the elements – or just the periodic table – is a table that displays all of the elements. What is the Periodic Table of the Elements? 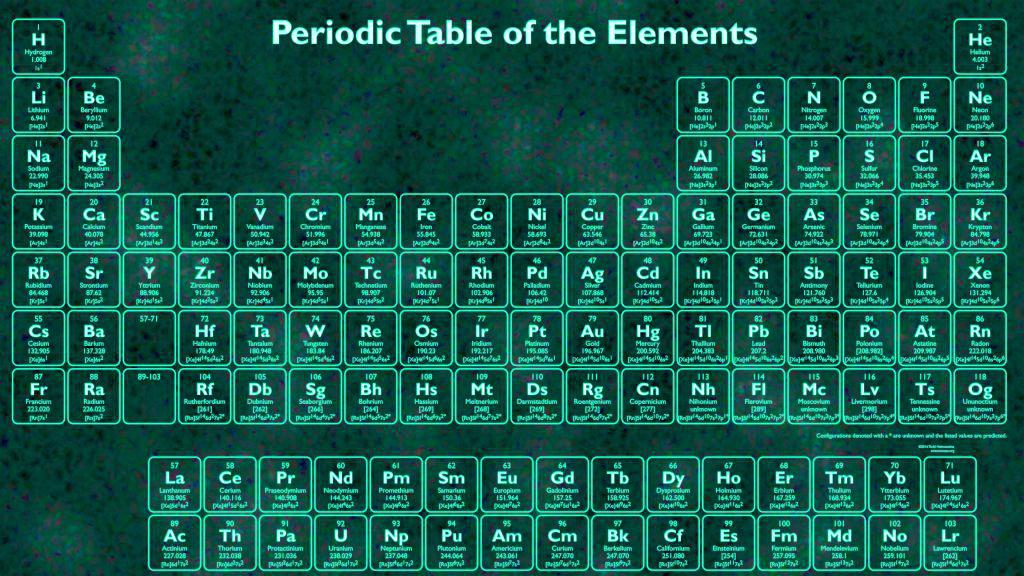
But first, let’s look at what’s in this overview article: In this guide, we’re going to bring some clarity to the periodic table for students (and parents). Introduction to The Beginners Guide to the Periodic Table To help you navigate the periodic table and learn about its history, we’ve produced this Beginner’s Guide to the Periodic Table to help High School students of all levels master the table of elements! When you think about it, there are 118 elements, classified into various categories according to their properties, and each element has a symbol and key attributes. Are you struggling to get your head around the periodic table? It’s a common issue for students.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
